Objective: To demonstrate the effects of water and CO2 on a heating atmosphere. To establish this experiment, we placed one temperature probe in a bottle under a heat lamp and another outside the heat lamp for 10 minutes, then moved the bottle out of the light and recorded data for 5 more minutes. For the second trial, we repeated the procedure, this time with 200 mL of water in the bottle. For the third trial, we repeated the procedure with 200 mL of water and CO2 from an alka seltzer tablet.
Note: How do i compile data???????
Raw data:
Note: How do i compile data???????
Raw data:
Conclusion questions:
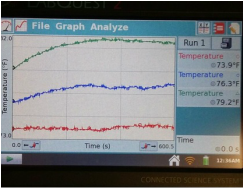
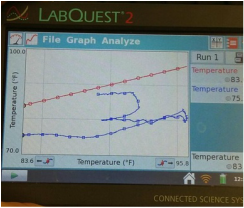
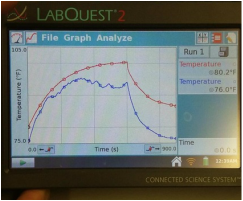
1. Describe the general characteristics of each graph.
For the first graph, each line followed about the same trend. For the second graph, the red and blue lines followed a similar trend, with the blue line increasing at a higher rate after about 90 seconds, and for the third graph, the red and blue curved sharply up and then leveled off, and then began to drop sharply around 600 seconds.
2. In which bottle did the temperature rise the greatest? Why do you think this occurred?
The temperature rose the highest in bottle two, where the red line rose from 85 to 100 degrees and the blue went from 75 to almost 90. I believe that water vapor bottle became the hottest because water vapor is a greenhouse gas that traps heat inside of the bottle. The CO2 bottle should have had high temperatures as well, but possibly something went wrong with our experiment, such as a leak in the bottle or a defect in the recording device.
3. Was there any difference
1. Describe the general characteristics of each graph.
For the first graph, each line followed about the same trend. For the second graph, the red and blue lines followed a similar trend, with the blue line increasing at a higher rate after about 90 seconds, and for the third graph, the red and blue curved sharply up and then leveled off, and then began to drop sharply around 600 seconds.
2. In which bottle did the temperature rise the greatest? Why do you think this occurred?
The temperature rose the highest in bottle two, where the red line rose from 85 to 100 degrees and the blue went from 75 to almost 90. I believe that water vapor bottle became the hottest because water vapor is a greenhouse gas that traps heat inside of the bottle. The CO2 bottle should have had high temperatures as well, but possibly something went wrong with our experiment, such as a leak in the bottle or a defect in the recording device.
3. Was there any difference
 RSS Feed
RSS Feed
